Radiolabeling Development.
From first Idea to Routine QC.
Radiolabeling is not just a chemical step – it defines the success of your entire development, validation and GMP process.
You want to establish robust radiolabeling processes and ensure reliable product quality? Even small variations in labeling and formulation can affect stability, efficacy and patient safety – and ultimately put your project at risk.
At CUP Contract Labs, we combine radiolabeling development with advanced analytics and microbiological testing in one integrated workflow.
This ensures data consistency, short turnaround times and reliable results – from early development to routine GMP quality control.
Where Radiolabeling becomes critical.
And why projects fail in real-world development.
High complexity, high pressure – and little room for error.
Radiolabeling of radiopharmaceuticals is inherently challenging. Short half-lives, sensitive chemistry and strict regulatory expectations create a development environment where even small deviations can have significant consequences.
Process conditions, precursor quality and analytical setup directly influence radiochemical purity, yield and stability. At the same time, increasing GMP requirements demand reproducibility, robustness and full process understanding.
Without a structured and integrated approach, these factors quickly lead to inefficiencies, failed transfers and unreliable results.
Why Projects fail in practice:
In real-world development, radiolabeling is highly sensitive to multiple variables:
- Radiolysis-driven degradation affects impurity profiles and product stability
- Precursor and formulation sensitivity influence yield and radiochemical purity
- Scale-up challenges: small-scale success does not guarantee clinical reproducibility
- Technology transfer risks between labs, CROs and CDMOs
- Analytical variability limits comparability and robustness
Most failures are not scientific – they are caused by insufficient process understanding and lack of control.
From Fragmentation
to Integration.
Radiolabeling development is often fragmented across partners, methods and data systems – creating inconsistencies, delays and risks during validation and technology transfer.
At CUP Contract Labs, we integrate radiolabeling, analytics and GMP requirements into one consistent workflow to ensure efficient development, reproducible results and a reliable transition into routine GMP quality control.
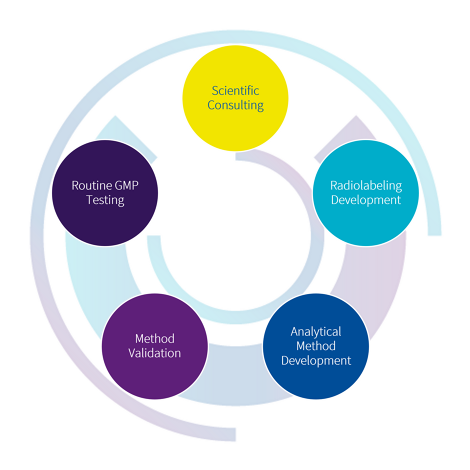
Our 6-Step Approach
to GMP Radiolabeling Development.
Analytical methods are the foundation of reliable radiolabeling development.
Core methods such as radio-TLC, LC-MS and ICP-MS enable the identification, quantification and characterization of your product – including efficiency, impurities, identity and stability.
From analytical methods to GMP-ready performance.
Ensuring reliability, reproducibility and compliance.
Analytical methods must not only generate data – they must deliver reliable and reproducible results under GMP conditions.
We systematically optimize and validate all methods to ensure:
- Clear separation and identification of all relevant compounds
- Reliable detection and quantification of impurities
- Consistent performance across systems and operators
- Stability of results under varying conditions
GMP Method Validation.
From development to validated performance.
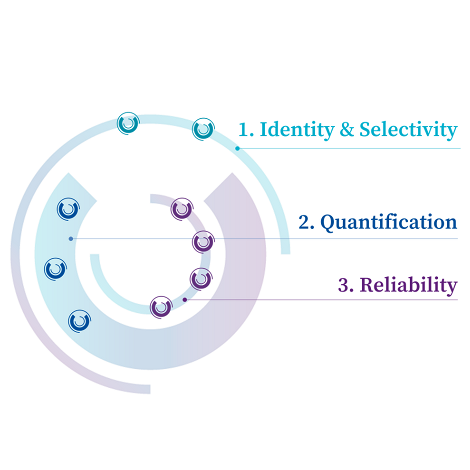
Method validation ensures that analytical methods are fit for GMP use, in accordance with ICH Q2 guidelines.
We follow a structured validation approach to receive GMP-compliant analytical methods ready for routine use and reliable release.
Regulatory Insight:
Validation Requirements in Clinical Development.
Validation requirements for analytical methods depend on the stage of clinical development and are often misunderstood in radiopharmaceutical projects.
For early-phase clinical trials, full validation is typically not required. However, the suitability of analytical methods must be clearly demonstrated, including defined parameters and acceptance criteria.
In later development phases, regulatory expectations increase. Analytical methods must be supported by structured validation data, usually provided in summarized form as part of the IMPD documentation.
While this guidance reflects EU IMPD requirements, similar principles apply to IND submissions in the US, where analytical method suitability and validation expectations are also critical components of CMC documentation.
Understanding these requirements early helps avoid unnecessary effort in early stages while ensuring a smooth transition into later-phase development and regulatory approval.
What drives successful
radiolabeling development.
From robust chemistry to reliable GMP performance.
Successful radiopharmaceutical development requires more than functional radiolabeling.
It requires:
- Robust radiolabeling chemistry
- Data-driven analytical development
- GMP-ready and validated methods
Our approach combines all of these elements in one integrated and structured workflow – from early development to routine quality control.
Result: Reliable and compliant batch release – every time.
expert voices
I am fascinated by radiolabelling because it combines radiochemistry and state-of-the-art analytics. I can see the results immediately, optimise methods and thus ensure that our radiopharmaceutical products meet the highest quality standards right from the start – for the protection of patients.



Let’s make your radiolabeling
GMP-ready.
We support you from first concept to routine quality control.
Our work is performed in GMP-compliant laboratories with specialized infrastructure for handling radioactive substances and complex pharmaceuticals.
This enables reliable results – even under the most demanding conditions.



